|
Background: There is a big contradiction in the medical literature regarding the relationship between atopy and contact hypersensitivity. Some researchers believe that atopy would prevent, whereas others believe that it would promote, the development of contact allergy. Possible causes of this confusion range from different study populations to different definitions of atopy. Objective: To evaluate the relationship between atopy and contact hypersensitivity in a well-defined general population sample using objectively measurable markers. Methods: I studied 135 randomly selected students from 5 vocational schools: 73 women and 62 men aged 18 to 19 years. The following atopy markers were tested: positive skin prick test results, positive Phadiatop test results, and total IgE levels greater than 120 kU/L. Contact hypersensitivity was detected by using patch tests. Statistical analyses included the Fisher exact test, the Mann-Whitney U test, and calculation of odds ratios. Results: At least 1 positive skin prick test result was found in 23.7% (95% confidence interval [CI], 16.5%-30.9%) of study participants, positive Phadiatop test results were found in 20.0% (95% CI, 13.3%-26.7%), and total IgE levels greater than 120 kU/L were found in 23.7% (95% CI, 16.5%-30.9%). Positive patch test reactions were found in 28.1% (95% CI, 20.6%-35.7%) of participants, most frequently to thimerosal (18.5%; 95% CI, 12.0%-25.1%) and nickel (9.6%; 95% CI, 4.6%-14.6%). For persons with atopy markers, odds ratios for contact hypersensitivity ranged from 1.0 to 3.2, the highest being for nickel hypersensitivity among those with total IgE levels greater than 120 kU/L. None of these relationships were statistically significant. Conclusion: Atopy and contact hypersensitivity are independent phenomena. |
Two things are astonishing when searching the literature on the possible relationship between atopy and contact hypersensitivity: the abundance of articles that deal with this topic and how contradictory their conclusions are. Table 1 provides a selection of conflicting statements on this topic. Among possible causes of this confusion may be different study groups and different definitions of atopy used in particular studies. The aim of this study was to reassess this relationship in a clearly defined sample from the general population using objective markers of atopy and contact hypersensitivity.
| Exact quote from the article | Case group | Control group | Description of the results |
|---|---|---|---|
| "Allergic contact dermatitis was less frequent among atopics than among non-atopics."1 | Personal history of AD, AR, or asthma | Negative history of atopic disease | ACD diagnosed in 40% of 935 atopics and in 51% of 3,472 nonatopics. |
| "...the frequency of contact allergy was similar in nonatopics (21%) as in atopics (22%)..."2 | Personal history of AD, AR, or asthma | Negative history of atopic disease | Patch test results were positive in 22% of 176 atopics and in 21% of 360 nonatopics. |
| "Atopy represents a predisposing factor for contact hypersensitivity."3 | Diagnosis of AD | Not applicable | 77% of 282 children with patch test-positive eczema were diagnosed as having AD. |
| "No association was found between contact allergy and atopic dermatitis or inhalant allergy."4 | Personal history of AD, AR, or asthma | Negative history of atopic disease | No significant association between contact allergy and atopy in the Mantel-Haenszel analysis of 1,146 children. |
| "Patch test in atopics is less frequently positive than in all patients with dermatitis."5 | Diagnosis of AD | Other forms of dermatitis | Largest difference observed was 2% positivity to PPD in patients with AD and 11% in the control group. |
| "Positive patch tests were significantly more frequent in atopic (28.8%) than in non-atopic (17.9%) children."6 | Personal history of AD, AR, or asthma | Negative history of atopic disease | Positive patch test results were found in 28.8% of 212 atopics and in 17.9% of 212 nonatopics. |
| "The frequency of positive reactions to patch testing with common contact allergens was found to be lower in patients with high IgE values."7 | IgE >1,000 U/mL | IgE <1,000 U/mL | Positive patch test results in none of 39 patients with high IgE levels compared with 15% in the control group. |
| "No correlation between serum IgE levels and contact sensitization."8 | Patch test results positive | Patch test results negative | Medium (124-999 U/mL), and high (>1,000 U/mL) IgE levels found, respectively, in 29% and 7% of 55 patch test-positive patients, and in 27% and 9% of 222 patch test-negative patients. |
| Abbreviations: ACD, allergic contact dermatitis; AD, atopic dermatitis; AR, allergic rhinitis; PPD, paraphenylenediamine. | |||
Study group
This prospective study was performed between February 25 and May 23, 2002. Random students from 5 vocational schools were invited to undergo tests for the presence of atopy and contact hypersensitivity markers. The number of participants needed for the study was calculated using the PS power and sample size calculations software.9 The α value (significance level) was set at .05, the β value was set at .20 (statistical power = .80), the nonatopic-atopic ratio was predicted to be 2.5, and the detectable difference in prevalence ratios of contact allergy was set at 20%. From these input data, the number of necessary observations was estimated to be 140. The vocational schools visited during the study were located in central, western, and southern Poland, at least 100 km apart. In each school, 1 class was randomly selected and invited to participate. The only criterion for inclusion was that at least 90% of the students in this class agreed to undergo the examinations. The study was performed on a voluntary basis. The study protocol and the manner of collecting informed consent were approved by the local ethics committee and the respective school authorities. Of 147 students who volunteered to participate in the study, 135 (91.8%) completed all the examinations. The study group consisted of 73 women and 62 men aged 18 to 19 years predominantly from rural areas (87.8%).
Measurement of the Frequency of Atopy and Contact Hypersensitivity
To avoid bias by either the participant or the researcher, only objectively measurable markers were used in this study. Positive skin prick test results, positive Phadiatop test results, and total IgE levels greater than 120 kU/L were used as atopy markers. Prick tests were performed using 15 aeroallergens considered to be relevant for this study group (Table 2).10 The test reactions were read after 15 minutes and were interpreted as positive if at least 1 allergen caused a wheal reaction of 3 mm or more in diameter.11-13 Phadiatop tests and determination of total IgE levels in the students' serum samples were performed using fluorescent enzyme immunoassay (FEIA, UniCAP 100, Pharmacia & Upjohn, Uppsala, Sweden). Contact hypersensitivity was detected using patch tests with the 10 contact allergens considered to be most frequent in this age group (Table 3).14 The allergen preparations (Chemotechnique, Malmö, Sweden) were applied on each participant's back using patch test units (IQ Chambers; Chemotechnique) and were removed 48 hours later. For logistic reasons (on-site examinations), the test reaction was read only once, 30 minutes after removing the patches. Reactions of at least "1+" (the presence of erythema and palpable infiltrate) were considered to be markers of contact hypersensitivity.15-17
Statistical Analysis
The frequencies of positive test results were calculated as percentages with 95% confidence intervals (CIs). Each of the 3 atopy markers (positive prick test results, positive Phadiatop test results, and total IgE levels > 120 kU/L) was separately cross-tabulated against patch test results split into 3 subcategories: any positive results, thimerosal-positive results, and nickel-positive results. Thimerosal and nickel were selected a posteriori as the most frequent individual sensitizers. The Fisher exact test was used to evaluate the significance of the relationships between atopy markers and contact hypersensitivity. In addition, the differences in total IgE levels between groups with and without contact hypersensitivity were analyzed using the Mann-Whitney U test. Odds ratios (ORs) with 95% CIs were calculated to evaluate whether the presence of atopy markers would increase (or decrease) the risk of contact hypersensitivity. Also analyzed was the relationship between female sex and the results of patch testing with nickel. This relationship is well-documented,18-20 and was regarded as a "positive control" for the study design. The statistical analyses were performed using software program (SPSS; SPSS Inc, Chicago, Ill).
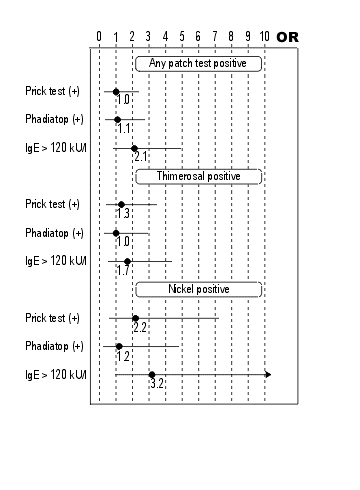
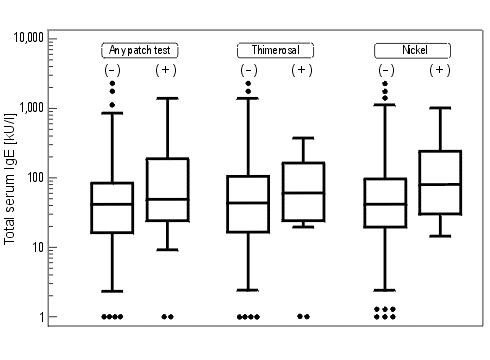
At least 1 positive prick test reaction was found in 23.7% (95% CI, 16.5%-30.9%) of study participants, positive Phadiatop test results were found in 20.0% (95% CI, 13.3%-26.7%), and total IgE levels greater than 120 kU/L were found in 23.7% (95% CI, 16.5%-30.9%). Detailed skin prick test results are given in Table 2. Positive patch test reactions were found in 28.1% (95% CI, 20.6%-35.7%) of study participants, most frequently to thimerosal and nickel (Table 3). A higher frequency of contact hypersensitivity (40.6%) was found in persons with IgE levels greater than 120 kU/L than among in those with lower IgE levels (24.3%) (Table 4). Nickel hypersensitivity was also more frequent in participants with positive skin prick test results (15.6% vs 7.7%). However, none of the differences seen in Table 4 were statistically significant (P > .05 for all). Figure 1 shows the ORs for contact hypersensitivity related to the presence of atopy markers. For particular markers, the ORs ranged from 1.0 to 3.2, the highest being for nickel allergy among persons with high IgE levels. None of these ORs were significant (P > .05 for all). In contrast, the "control" OR for nickel allergy among women was significant (OR, =12.0; 95% CI, 1.5-95.2; P = .003). ). Median IgE levels were slightly higher among individuals with positive patch test results; however, these differences were not significant (Mann-Whitney U test: P = .27, .43, and .12 for any positive results, thimerosal-positive results, and nickel-positive results, respectively).
| Allergen | Positive results, % (95% CI) |
|---|---|
| Lepidoglyphus destructor* | 12.6 (7.0-18.2) |
| Acarus siro* | 11.1 (5.8-16.4) |
| Dermatophagoides pteronyssinus* | 10.4 (5.2-15.5) |
| Grain dust | 6.7 (2.5-10.9) |
| Hay dust | 5.9 (1.9-9.9) |
| Tyrophagus putrescentiae* | 4.4 (1.0-7.9) |
| Weed pollen* | 4.4 (1.0-7.9) |
| Grass/cereals pollen* | 3.0 (0.1-5.8) |
| Straw dust | 3.0 (0.1-5.8) |
| Tree pollen I* | 2.2 (0.0-4.7) |
| Tree pollen II* | 1.5 (0.0-3.5) |
| Animal dander I* | 0.7 (0.0-2.2) |
| Cow epithelium* | 0.7 (0.0-2.2) |
| Pig epithelium* | 0 |
| Horse epithelium* | 0 |
| Abbreviations: CI, confidence interval
*Allergens are from Allergopharma (Reinbek, Germany). Allergens are from Biomed (Krakow, Poland). |
|
| Allergen | Positive results, % (95% CI) |
|---|---|
| 0.1% Thimerosal | 18.5 (12.0-25.1) |
| 2.5% Nickel sulfate | 9.6 (4.6-14.6) |
| 1% Cobalt chloride | 6.7 (2.5-10.9) |
| 0.5% Potassium dichromate | 3.0 (0.1-5.8) |
| 8.0% Fragrance mix | 0.7 (0.0-2.2) |
| 0.1% Mercuric chloride | 0 |
| 20% Neomycin sulfate | 0 |
| 1% Mercapto mix | 0 |
| 0.6% Black rubber mix | 0 |
| 25% Balsam Peru | 0 |
| Abbreviations: CI, confidence interval. | |
| Atopy marker | Any positive results, % | Thimerosal-positive results, % | Nickel-positive results, % |
|---|---|---|---|
| Skin prick test results | |||
| Positive | 28.1 | 21.9 | 15.6 |
| Negative | 28.1 | 17.5 | 7.7 |
| Phadiatop test results | |||
| Positive | 29.6 | 18.5 | 11.1 |
| Negative | 27.8 | 18.5 | 9.2 |
| Total IgE, kU/L | |||
| >120 | 40.6 | 25.0 | 18.7 |
| <120 | 24.3 | 16.5 | 6.8 |
| *No statistically significant differences were found for any of the measures (P > .05). | |||
Figure 1. Relationship between the presence of atopy markers and contact hypersensitivity. Data are given as odds ratios (ORs), with the bars representing 95% confidence intervals.

Figure 2. Total IgE levels in participants with positive vs negative patch test results. "Whiskers" represent ranges of measured values, boxes represent interquartile ranges, horizontal bars within boxes are the medians. The individual dots represent outliers.
The scientific question of the present study was: What is the relationship between atopy and contact hypersensitivity? For a long time, our understanding of this problem was influenced by the widely accepted concept of a counterbalance between atopy (type I allergy) and contact hypersensitivity (type IV allergy). This theoretical concept, discussed in detail elsewhere,8,21 was supported by in vitro studies that showed impaired cellular responsiveness in atopic patients. Despite this finding, data from clinical studies remained contradictory (Table 1). Possible reasons for the conflicting results range from different study groups to remarkable differences in definitions of atopy. Additional confusion was due to the fact that generalized conclusions about atopy were sometimes based merely on observations of atopic eczema.1,3,5
The frequency of atopy in the present study group is slightly lower than that in previously studied random Polish populations. In a group of farming students, prick test reactions were positive in 30.9% (vs 23.7% in the present study), Phadiatop test results were positive in 32.3% (vs 20.0%), and total IgE levels were greater than 120 kU/L in 34.6% (vs 23.7%).10 Also, in a representative sample of Polish rural residents, positive prick test reactions to aeroallergens were found in 28.6%.22 On the other hand, the rate of contact hypersensitivity was higher in the present study than in a cohort of Polish schoolchildren aged 13 to 15 years,19 among whom 21.6% had positive patch test results (vs 28.1% in the present study).
This study was performed in schools with up to 30 students each tested within a few hours, which imposed certain methodological limitations. Prick tests were restricted to 15 allergens, whereas a broader array might possibly increase the number of students with positive results. Patch tests were restricted to 10 haptens and were read only once after 2 days, whereas an additional reading after 5 to 7 days might affect the final positivity rates.23,24 Such readings, however, were impossible because of the long distances between the schools and the predicted unwillingness of students to participate in a study that would last longer than 2 days. This assumption seems to be confirmed by the fact that 12 students did not participate in the first patch test reading (dropout rate, 8.2%). Despite the initial excess of participants, the final number of students who completed the study (n = 135) was lower by 3.6% than the initially planned 140. Among the strengths of the study, the exclusion of interobserver variability deserves mention, as 1 physician performed all the tests. Only objectively measurable test results were analyzed to minimize the subjective bias. The study population was highly homogenous regarding age and environmental exposures - factors that affect prick and patch test results.20,22 Finally, the relationship between contact allergy and atopy was, for the first time, addressed by using the Phadiatop test, regarded as an objective atopy detection method and recommended for standardizing epidemiologic studies.25-29
To my knowledge, only 2 studies on the relationship between atopy and contact hypersensitivity have previously been performed in samples from the general population. To define the presence of atopy, history of atopic disease was used in 1 study,4 whereas positive prick test results and elevated total IgE levels were used in the other.30 Results from both studies suggested no association between atopy and contact sensitization. The previous observations are reinforced by the present study results, which are based exclusively on objective measures, including 3 various atopy markers. In conclusion, atopy and contact hypersensitivity seem to be independent in the general population.
I thank Agnieszka Drwal, RN, for her assistance in prick and patch testing, Nimfa M. Stojek, PhD, for performing the IgE measurements, and Susan Gibbs, PhD, for checking the manuscript's English style. This study was supported by grant KBN 6 P05D 028 20 from the State Committee for Scientific Research, Warsaw, Poland.
For personal use only. © Annals of Allergy Asthma Immunology
| Contact Dr. Spiewak | Back to article list | Website's front page |