|
The authors present the case of a carpenter with bronchoconstriction after exposure to the wood dust of trees common in Central Europe. Ash wood dust was the most potent bronchoconstrictive agent. The patient's medical history and the results of bronchial provocation suggested occupational, non-atopic asthma. Key words: occupational asthma, wood dust, ash, oak, beech, pine, bronchial provocation tests. |
Patient K.Z., a carpenter, aged 43, was admitted to the Outpatient Allergy Clinic in July 1993 with unconfirmed bronchospasms due to wood dust exposure at his workplace. For over 20 years he had been regularly exposed to wood dust while sawing, filing and polishing wood. The first episodes of expiratory dyspnoea occurred in 1986 (after 15 years of work as a carpenter) and came repeatedly two to three times a week. The onset or increase in dyspnoea took place during work in a dusty environment and continued for several hours after work had finished. During these episodes he also suffered from eye watering and rhinitis. The symptoms increased gradually. After 4 years the dyspnoea became persistent, but only increased after exposure to wood dust at the workplace. There were four kinds of wood used at the carpenter's work: ash, oak, beech and pine. The patient was not able to indicate which of the woods mentioned above were responsible for his problems. The symptoms increased from Monday to Friday, and were more severe by the end of work. The patient's condition used to improve partially after several days' cessation of work. In 1991, after a considerable increase in the symptoms, the patient was hospitalized in the Pulmonary Unit where, on the basis of microbiologic tests and radiograms, tuberculosis was diagnosed. The patient's return to work, after 9 months of treatment, was connected with the return and gradual increase in dyspnoea. An increase in his symptoms resulted in his next hospitalization in the Pulmonary Unit. This time active tbc was excluded based on the absence of mycobacteria in his sputum and no changes on radiograms suggesting fibrotic involvement of the lung. After this hospitalization the patient did not return to work and his health began to improve significantly.
Past history: infantile eczema in childhood. The patient has smoked twelve cigarettes a day since 1971. Family anamnesis: the patient's daughters manifest pollinosis symptoms. Examination on the first visit: body weight 60 kg, height 165 cm, blood pressure of 125/80 mmHg, pulse of 80 per min., respiration of 18 breaths per min. and no physical abnormalities found. Laboratory data: Hb - 9.37 mmol/l, Hct - 0.42, ESR - 11 mm/h, RBC - 4.73 T/l, WBC - 7.6 G/l, neutrophils - 45%, eosinophils - 3%, basophils - 1%, lymphocytes - 49%, monocytes - 2%, total serum IgE - 273 IU/l. Spirometry: FEV1 - 3.20 l (95.18 % of reference value), FVC - 3.60 l (89.05%), MEF25 - 2.05 l/s (110.78%), MEF50 - 4.17 l/s (91.29%), MEF75 - 7.00 l/s (96.06%), PEF - 7.42 l/s (88.02%), VC - 3.82 l (90.66%), IC - 2.25 l (76.77%), ERV - 1.57 l (122.44%), MVV - 108.69 (87.76%). Blood gases: pO2 - 11.8 kPa, pCO2 - 5.3 kPa, pH - 7.38, HCO3- - 22.9 mmol/l.
Standard allergologic tests: skin prick tests revealed hypersensitivity to Dermatophagoides pteronyssinus (wheal 7 mm/erythema 35 mm), Dermatophagoides farinae (7/30) (Allergopharma), house dust (7/30), straw dust (6/16), cereal dust (6/30), hay dust (7/30) and Candida albicans (5/20) (Biomed) at a control reaction to histamine of (6/20) and to 0.9% NaCl of (0/9). Due to suspicion of non-atopic occupational asthma caused by hypersensitivity to the wood dust the patient was exposed to at his workplace, bronchial challenge tests were carried out.
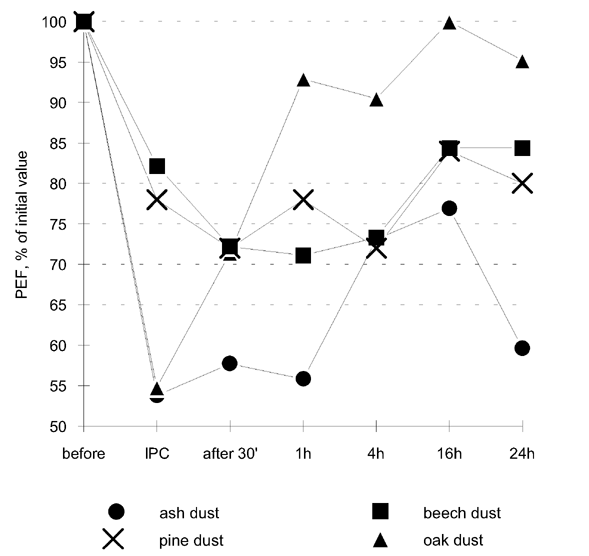
Wood dust bronchial provocation tests were conducted by having the patient stay for half an hour in conditions similar to those at his workplace. All the kinds of wood used by this carpenter at his work were tested with 1 week intervals in order to avoid an overlap of allergen-induced bronchial hyperreactivity. The wood dust of the following trees was used: ash (Fraxinus excelsior), oak (Quercus sp.), beech (Fagus silvatica) and pine (Pinus sylvestris). For each test the wood dust of the tree being tested was raised mechanically in a 2 m3 test chamber. The mean wood dust concentration in the air breathed was approximately 2-3 mg/m3 . The results of these bronchial provocation tests are shown in Table 1 and Figure 1.
| before | IPC | 30' | 1h | 4h | 16h | 24h | |
|---|---|---|---|---|---|---|---|
| Ash dust | 520 | 280 | 300 | 290 | 380 | 400 | 310 |
| Ash extract | 540 | 310 | 290 | 300 | 310 | 380 | 400 |
| Oak dust | 420 | 230 | 300 | 390 | 380 | 420 | 400 |
| Oak extract | 450 | 320 | 350 | 350 | 310 | 400 | 420 |
| Beech dust | 450 | 370 | 325 | 320 | 330 | 380 | 380 |
| Beech extract | 400 | 320 | 290 | 300 | 290 | 370 | 370 |
| Pine dust | 500 | 390 | 360 | 390 | 360 | 420 | 400 |
| Pine extract | 450 | 380 | 390 | 400 | 370 | 390 | 390 |
| Distilled water | 380 | 290 | 340 | 360 | 360 | 340 | 360 |
| 0.9% NaCl | 400 | 380 | 400 | 380 | 390 | 400 | 390 |
| IPC - immediately post challenge | |||||||
Figure 1. The results of wood dust provocation tests. IPC - immediately post challenge.

Next, to limit the possibilities of nonspecific irritant bronchoconstriction due to the inhalation of wood dust particles, bronchial provocation tests with aqueous wood extracts were performed. The wood dust was mixed with 0.9% NaCl in a 1:1 proportion. The suspension was kept at room temperature for 48 hours, shaking when necessary; then, the clear supernatant was taken and after being filtered through a bacterial filter was stored at 4°C.
These aqueous solutions were used for prick tests which produced the following results: histamine (4/20), 0.9% NaCl (0/0), ash (0/0), oak (0/0), beech (0/0), pine (0/6). In addition there were no late phase skin reactions. After this, bronchial provocation tests based on inhalation for 30 minutes of these solutions, diluted to 1:4 (v/v), were performed using an ultrasonic inhalator (Thomex L2, Warsaw). Spirometry was conducted before and immediately after the provocation, as well as at 30 minutes and 1, 4, 16 and 24 hours after the end of the inhalation. The results obtained were compared with the bronchial challenge results carried out with distilled water as a factor causing nonspecific bronchoconstriction and with 0.9% NaCl. The results are shown in Table 1 and Figure 2.
Figure 2. The results of bronchial provocation tests with aqueous wood extracts, 0.9% NaCl and distilled water. IPC - immediately post challenge.
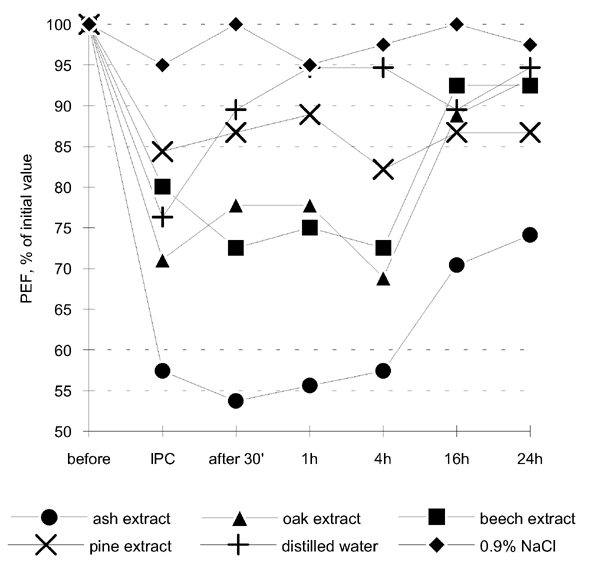
At first sight, the decrease in PEF values is most visible for ash wood both as a dust and as an extract. The next step was to test the wood dust and aqueous solutions used in the provocations for bacterial and fungal contamination in order to exclude microorganism antigens as a cause of immunologically mediated bronchoconstriction. Microbiological cultures were made on the blood agar, MacConkey agar, Universal Medium No 2 and Löwenstein-Jensen medium with negative results. The precipitation test using Ouchterlony technique with the patient's serum and the wood extracts described above was also done with negative result.
Cases of pneumopathies caused by inhaling the wood dust of certain trees are discussed in the literature [6, 7, 8, 10, 11]. Sosman et al. [10], reporting hyper-sensitivity reactions to mahogany and Western red cedar wood dust, stress that they can cause either an asthmatic response or extrinsic alveolitis. Analysis of serum from carpenters with bronchospasm caused by exposure to exotic tree wood dust revealed the presence of precipitins reacting with these wood extracts [10]. The case of a carpenter with diffuse infiltrative fibrosis and the presence of granuloma in a pulmonary biopsy, which were explained as reactions to exotic tree wood dust, has also been described [6]. In the 1970s and early 1980s reports about occupational asthma due to exposure to Western red cedar appeared [1, 2, 3, 4, 7]. Cockcroft et al. [5] suggest that the bronchoconstriction after exposure to wood dust is caused by low molecular chemicals (plicatic acid, abietic acid) of uncertain action mechanisms. These chemicals are said to be the most common sensitizing agents in woods.
In the literature [9] there are reported cases of the supposed role of bacterial and mycologic antigens in the pathogenesis of pneumopathies caused by wood dust exposure. In the case presented in this paper this was excluded by thorough microbiological tests. Previous tuberculosis, due to the known adjuvant role of Mycobacterium antigens, could have played a role in the development of the patient's present disorders. However, his respiratory problems began about 5 years before tbc was first diagnosed. After careful analysis of the patient's history (the latent period, immediate and late symptoms and history of "crescendo" symptoms when exposed to wood dust) and of the bronchial challenge results (diphasic bronchoconstriction with a prolonged late phase), occupational non-atopic asthma due to wood dust (ash, oak, beech and pine) has been diagnosed with great probability [5, 11]. The negative results of the skin tests on the patient suggest the absence of IgE-mediated reactions. Diffuse pathologic processes, such as extrinsic allergic alveolitis can be excluded based on the following facts: there were no fibrotic changes on the chest radiograms, no functional changes in terminal airways, no signs of disturbed pulmonary gas exchange, no systemic reactions (e.g. fever, tachycardia) after being challenged, and the blood leukocytosis was within a normal range. Also, normal FVC and bronchial hyperreactivity to nonspecific stimuli such as the inhalation of distilled water make the diagnosis of allergic alveolitis less probable [11]. The negative result of the Ouchterlony test with the wood extracts under examination seems to exclude the pathogenic role of serum precipitins. Possibly the cause of bronchoconstriction in the patient are the low molecular chemicals mentioned above.
The case presented here seems to be one of the very few descriptions that have been published till now of asthma caused by exposure to the wood dust of trees common in Central Europe (a comprehensive summary of the literature can be found in [11]). To the best of our knowledge this is the first documented case of hypersensitivity to ash (Fraxinus excelsior). In our opinion, broncho- and pneumopathies caused by exposure to wood dust are underdiagnosed and we hope that in the near future this problem will be reanalysed on a greater number of patients.
For personal use only. © Radoslaw Spiewak
| Contact Dr. Spiewak | Back to article list | Website's front page |